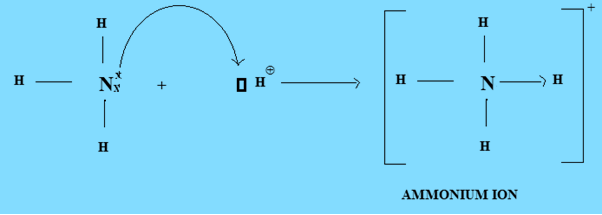
The displayed formula shows all bonds and all atoms in the structure. We’re being asked to identify the displayed formula of the ammonium ion in this question. Place the remaining two electrons on the nitrogen atom. In the ammonium ion, which has the formula NH4 , nitrogen is the donor atom as far as the formation of the coordinate covalent bond is concerned. Now draw a third hydrogen atom next to the nitrogen atom and place a third pair of electrons between these atoms. Now draw a second hydrogen atom next to the nitrogen atom and place a second pair of electrons between these atoms. The total number of electrons for ammonia will therefore be 8 electrons.ĭraw a hydrogen atom next to a nitrogen atom and place a pair of electrons between these two atoms. The nitrogen atom has 5 electrons and each of the three hydrogen atoms has 1 electron. A bond in which one atom has donated both electrons is. What are the numbers of bonding electrons for the nitrogen atom and the hydrogen atoms? When the hydrogen ion bonds to the ammonia molecule, both electrons of the bond come from the nitrogen. The arabic numeral above the element's column in the periodic table gives you that number. In short, it is a bond formed by the attraction between two oppositely charged ions. It is formed by the protonation of ammonia ( NH 3). The two lone pairs are on opposite sides of the octahedron (180° apart), giving a square planar molecular structure that minimizes lone pair-lone pair repulsions ( Figure 7.19 ). Its overall electrical charge is zero, and its atoms are held together by both covalent bonds and an ionic bond. When a central atom has two lone electron pairs and four bonding regions, we have an octahedral electron-pair geometry. The first step in drawing the electron dot formula for ammonia is to determine the number of bonding electrons for each of the atoms. IONIC BOND An ionic bond is a type of chemical bond that involves a metal and a non-metal ion (or polyatomic ions such as ammonium) through electrostatic attraction. The ammonium cation is a positively charged polyatomic ion with the chemical formula NH 4 or NH 4 . Ammonium chloride (NH4 Cl-) is a molecule. What is the first step that you must do in order to draw the electron dot formula for ammonia, NH 3? If you get stuck, try asking another group for help. You should try to answer the questions without referring to your textbook. The modification occurs after the amino acid has been assembled into a protein.\) The hydrogen's electron is left behind on the chlorine to form a negative chloride ion. In some cases an amino acid found in a protein is actually a derivative of one of the common 20 amino acids (one such derivative is hydroxyproline). When the ammonium ion, NH4 , is formed, the fourth hydrogen is attached by a Coordinate covalent bond - Wikipedia, because only the hydrogen's nucleus is transferred from the chlorine to the nitrogen. Glycine, the major amino acid found in gelatin, was named for its sweet taste (Greek glykys, meaning “sweet”). It was obtained from protein found in asparagus juice (hence the name). Together, they comprise a single ion with a 1 charge and a formula of NH 4 . The first amino acid to be isolated was asparagine in 1806. The ammonium ion (see figure below) consists of one nitrogen atom and four hydrogen atoms. The only amino acid whose R group has a pK a (6.0) near physiological pHĪlmost as strong a base as sodium hydroxide Oxidation of two cysteine molecules yields cystineĪmino acids with a negatively charged R groupĬarboxyl groups are ionized at physiological pH also known as aspartateĬarboxyl groups are ionized at physiological pH also known as glutamateĪmino acids with a positively charged R group On the other hand, ammonium ion has a positive. Named for its similarity to the sugar threose Ammonia is a neutral gas with chemical formula NH 3 i.e., it contains 3 hydrogen atoms and one nitrogen atom. Side chain functions as a methyl group donorĬontains a secondary amine group referred to as an α-imino acidĪmino acids with a polar but neutral R group The only amino acid lacking a chiral carbonĪn essential amino acid because most animals cannot synthesize branched-chain amino acidsĪlso classified as an aromatic amino acid \): Common Amino Acids Found in Proteins Common Name
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
